Hangzhou Clongene Biotech Co Ltd
Hangzhou Clongene Biotech Co.,Ltd. is established in 2004 with a registered capital of 50.1 million RMB. Area scales to 19,000 square meters in R&D and manufacturing base with about 2700m2 100-thousand-grade Purification Workshop, which is located in No.1 Yichuang Road, Yuhang Sub-district, Yuhang District, Hangzhou, China. Clongene is a high-tech biotechnology company in manufacturing biology raw materials, the second and third class of 6840 in vitro diagnostic tests, food safety tests, veterinary diagnostic tests, etc.
World Certifications & Recognitions
Clongene obtains several certifications and honors: ISO13485, ISO9001 certification & European Union CE certification; The U.S. FDA 510K marketing permit; 27 items China CFDA marketing approval; Recommend enterprise of drug abuse testing device in Ministry of Public Security; Put on record in China Ministry of Agriculture on food safety test device; Be selected as one of Military procurement suppliers; Evaluated as a SMB Technology Enterprise in Zhejiang province; Hangzhou R&D center; AAA-Credit enterprise.
Lungene® Antigen Test Cassette
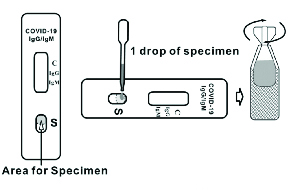
The COVID-19 Antigen Rapid Test Cassette is a lateral flow immunoassay intended for the qualitative detection SARS-CoV-2 nucleocapsid antigens in nasopharyngeal swab and oropharyngeal swab from individuals who are suspected of COVID-19 by their healthcare provider.
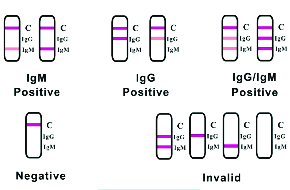
The FDA is allowing Clungene® SARS-CoV-2 IgG/IgM Rapid Serology Test to be made available under its new policy dated May 4, 2020. (Policy for Coronavirus Disease-2019 Tests During the Public Health Emergency). As required, the completed EUA template and associated documentation were submitted on May 15, 2020 to the FDA. The submission (EUA#201121) included updated validation reports and additional clinical data. Clungene's establishment registration & device number on the FDA's website is 3009414546.